Pickadook/Shutterstock
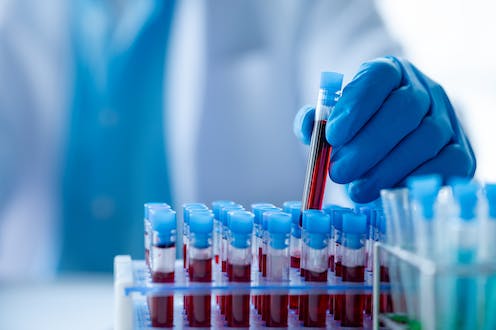
A five-year, £5 million initiative has just been launched to investigate the feasibility of using new blood tests in the diagnosis of Alzheimer’s disease. Alzheimer’s Research UK and the Alzheimer’s Society are working with the National Institute for Health and Care Research to use these blood tests in the NHS.
This funding, hot on the heels of recent drug trials for Alzheimer’s disease, continues a wave of breathless excitement in a field that has traditionally found good news stories hard to come by.
Of those seen by specialists in memory services, the vast majority are given a diagnosis of dementia based on their symptoms alongside cognitive tests, blood tests to rule out other explanations (such as hormone imbalances), and sometimes an MRI brain scan.
A small percentage, particularly those who are younger or who have more complex symptoms, may be offered a more detailed investigation to look for some proteins (amyloid and tau) that can build up in the brain.
At the moment, this would involve a lumbar puncture (placing a needle into the spine and removing some of the fluid) or a specialist brain scan called a PET scan. If simple blood tests can tell us the same information, with enough accuracy, then this will be preferable for this small group of people.
So far, so good. But what about those people who don’t need a lumbar puncture or PET scan? Will they see meaningful benefits from these new blood tests? It is far from certain.
Some argue that more “precision” in the diagnosis will help people understand what the coming years will entail. But this assumes that we can confidently place all people with dementia into the various disease categories (such as Alzheimer’s, vascular dementia, Lewy body dementia, frontotemporal dementia) based on the pathologies we find in their brains, and that we can then accurately predict how things will unfold for that person.
Unfortunately, we can’t. Instead, data show that many pathologies (disease-causing abnormalities such as protein build-up or damage to blood vessels) are linked to dementia, and most people with dementia have more than one. This mixed disease is a big part of what makes researching the syndrome of dementia so complex.
The other suggested benefit is that the tests will tell us if the patient is eligible for the new drugs approved in the US (and currently being considered by UK and European regulators). But beyond the headlines, the current crop of new drugs don’t stand up to scrutiny.
Beyond the amyloid theory
The theory on which they are based (that the build-up of the amyloid protein is the trigger for everything that comes after) is increasingly challenged by experts. We need to better understand the complex biology of dementia.
In the past, this has been heavily focused on amyloid even though evidence has told us to also look elsewhere.
The ultimate frontier that many seek is screening people who have no symptoms but who would, if tested, be found to have raised protein levels. They hope that by detecting people at this stage, drugs could not just slow down the disease but prevent it altogether.

Kittipong Somklang/Shutterstock
A recent trial tested this, in which people with raised amyloid but no symptoms took the amyloid-clearing drug solazenumab for more than four years. It had no effect on cognitive function compared with a placebo, confirming that this ambition is, sadly, not close.
It may not ever be realised for such a complex disease. The most significant problem is that many of those who have raised brain amyloid but no symptoms will die without developing dementia. Therefore, most symptom-free people with a positive blood test have nothing to gain and can only experience harm – from anxiety, further tests, or treatments. Importantly, the focus of this new initiative is not people without symptoms.
New initiative
Several studies of new blood tests have been carried out already in people with dementia symptoms, showing they are almost as good as PET scans or lumbar punctures at detecting protein levels. But the people in these studies were typically younger (in their 60s and 70s), with minimal brain pathologies (except amyloid) and other disorders, and minimal ethnic and socioeconomic diversity.
This initiative will test how well these emerging blood tests perform for those with suspected dementia in the NHS. Most people in the UK who develop dementia are in their 80s, have mixed brain pathologies, commonly have other diseases (for example, kidney disease, which may affect the accuracy of the blood tests, and rates are higher among poorer groups and those from some minority ethnic backgrounds.
The results of this new initiative will tell us how well the new blood tests perform in these older, more complex people. The most important question, though, will be: do the results of protein tests change the way we look after people with dementia, resulting in a better quality of life?
![]()
Sebastian Walsh receives funding from the NIHR
Edo Richard receives funding from The Netherlands Organisation for Health Research and Development (ZonMW) – public funding
Carol Brayne does not work for, consult, own shares in or receive funding from any company or organisation that would benefit from this article, and has disclosed no relevant affiliations beyond their academic appointment.



























